Panacea Biotec and RDIF to Produce 100 Million Sputnik V vaccine Doses Yearly
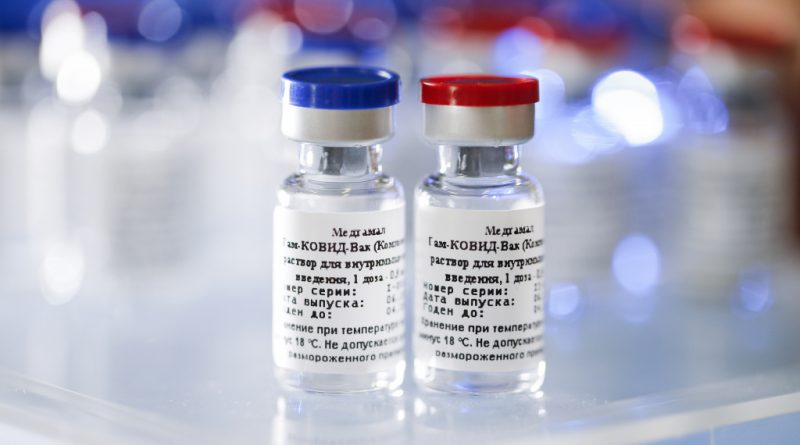
The Russian Direct Investment Fund (RDIF, Russia’s sovereign wealth fund), and Panacea Biotec, India’s largest vaccine and pharmaceutical manufacturer, have announced cooperation and formed a partnership to produce 100 million doses each year of Sputnik V, the world’s first recorded coronavirus vaccine.
100 Million doses of Sputnik V to be produced each year by Panacea Biotec
Sputnik V manufacturing at Panacea Biotec plants would aid in the global supply of Sputnik V to RDIF’s foreign partners. The Sputnik V COVID-19 vaccine has been registered in 59 countries around the world, with a combined population of more than 1.5 billion people. The vaccine is currently under rolling review by the European Medicines Agency (EMA). On Thursday, Germany’s health minister Jens Spahn announced that his country would engage in talks with Russia for a potential purchase of the vaccine.
Panacea Biotec has a strong track record of supplying more than 10 billion doses of Polio vaccines and over 150 million doses of Innovative fully liquid Vaccines to National Governments, UN Agencies, etc. Panacea Biotec’s vaccine manufacturing facilities are WHO Prequalified, pharmaceutical and sterile cytotoxic injectable facilities are USFDA approved.
About the Sputnik V COVID-19 vaccine
Sputnik V has a 91.6 % efficacy rate, according to reports released in the Lancet, one of the world’s oldest and most prestigious medical journals. The vaccine technology is established on a well-studied platform of human adenoviral vectors, and it uses two distinct vectors for each of the two shots in a series of vaccination, resulting in longer-lasting immunity than vaccinations that use the same delivery system for both shots. Sputnik V vaccine researchers are collaborating with AstraZeneca on a joint clinical trial to enhance the effectiveness of the AstraZeneca vaccine.
Kirill Dmitriev, CEO of the Russian Direct Investment Fund, commented:
“Vaccine partnerships are the only way to overcome the pandemic. The world continues its fight against coronavirus and we see a growing interest in Sputnik V vaccine as it is one of the best vaccines available. Cooperation with Panacea Biotec is an important step to produce the vaccine in India and to supply our international partners around the world.”
Dr. Rajesh Jain, Managing Director of Panacea Biotec, said:
“We are pleased to collaborate with RDIF to produce Sputnik V for global markets. Panacea Biotec brings decades of vaccine manufacturing and distribution know-how to scale-up Sputnik V supplies. Panacea Biotec will produce Sputnik V in its internationally accredited facilities complying to strict GMP standards and prequalified by WHO.”
91.6% efficacy against severe COVID-19 complications
Sputnik V has many advantages including that it has a confirmed 91.6% efficacy providing full protection against severe COVID-19 complications. Furthermore, the Sputnik V vaccine technology is a well-studied and validated platform of human adenoviral vectors, which have been around for thousands of years well known to cause the common cold.
Within the last two decades, more than 250 scientific trials have shown the reliability, effectiveness, and absence of long-term side effects of adenoviral vaccines. In this regard, it is confirmed that there are no strong allergies caused by Sputnik V. Also it provides long-lasting immunity due to the fact that Sputnik V uses two separate vectors for each of the two shots in a vaccination sequence versus vaccinations that use the same delivery mechanism for both shots.
Sputnik V costs less than $10 per shot and can be stored at standard temperatures
In addition to this, Sputnik V is cost-effective and easy for distribution and store globally. Taking into consideration that the price of Sputnik V is less than $10 per shot and the storage temperature of Sputnik V is +2 to +8 °C which is within the range of standard refrigerators, additional cold chain logistic expenses can be eliminated, making the vaccine accessible worldwide.
Recommended Companies
Ad
More Headlines