Chiesi and Protalix’s Therapy for Fabry Disease Receives Marketing Approval from EC
Chiesi Global Rare Diseases and Protalix BioTherapeutics, Inc. have received marketing authorization from the European Commission (EC) for their drug, PRX-102 (pegunigalsidase alfa), in the European Union (EU). The drug is indicated for the treatment of adult patients suffering from Fabry disease.
PRX-102 is a recombinant human α‑Galactosidase‑A enzyme expressed through ProCellEx® plant cell-based expression system
PRX-102 is a PEGylated enzyme replacement therapy (ERT). It is a recombinant human α‑Galactosidase‑A enzyme expressed in plant-cell culture that is designed to provide a long half-life. The EC granted marketing authorization for PRX-102, based on a thorough clinical development program involving over 140 patients who received up to 7.5 years of treatment.
Chiesi Global Rare Diseases, which is a business unit of the Chiesi Group dedicated to developing innovative therapies for rare diseases, collaborated with Protalix BioTherapeutics, a biopharmaceutical company focused on the commercialisation of recombinant therapeutic proteins. The drug is expressed through the company’s proprietary plant cell-based expression system, ProCellEx®. The EC’s approval marks a significant milestone for individuals affected by Fabry disease, who may benefit from this new treatment option.
Giacomo Chiesi, head of Chiesi Global Rare Diseases said:
“People living with Fabry disease often perceive their disease as burdensome and still experience unmet medical needs. Our deepest gratitude to all patients and patient advocates who have stood shoulder-to-shoulder with clinical researchers, scientists and regulators during the clinical development program, providing the data needed for this approval. I believe this is a vital ingredient in bringing innovation to the real lives of patients and enabling hope and definitive, integrated solutions.”
Diego Ardigò, M.D., Ph.D., head of research and development of Global Rare Diseases at the Chiesi Group said:
“We are delighted that the European Commission has approved PRX-102 for the treatment of adult patients with Fabry disease. The EU authorization is a testament to our commitment to deliver innovative therapies and solutions for people affected by rare diseases. As a certified B Corp we are committed to ensuring access to PRX-102 to as many people living with Fabry disease as possible and thank those who participated in our extensive clinical research program. It is important to deliver this new treatment option to reduce the burden of this chronic disease on patients, their families, and the healthcare system.”
Dror Bashan, Protalix’s President and Chief Executive Officer stated:
“The European Commission’s approval of PRX-102 is a significant milestone for patients with Fabry disease and their families, providing a new therapeutic option. We are proud of this achievement and believe that this approval further validates our science and technology. Based on solid results from our robust clinical programs, PRX-102 has the potential to be widely used for many years to come. Together with Chiesi, we remain committed to meeting the needs of patients with Fabry disease and bringing this new treatment option to market.”
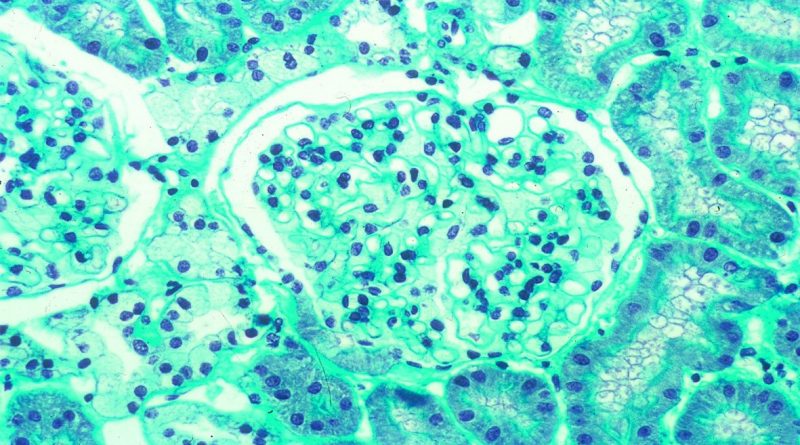
PRX-102 has been studied in both ERT-naïve and ERT-experienced patients, including a head-to-head trial that met its primary endpoint, with PRX-102 demonstrating non-inferior efficacy to agalsidase beta in controlling kidney disease as evaluated by the estimated glomerular filtration rate (eGFR) decline.
Pegunigalsidase alfa, an investigational new drug product, is currently not approved by the U.S. Food and Drug Administration (FDA). The effectiveness and safety of pegunigalsidase alfa is under review, but has not yet been approved, by the FDA.
Original Source – Press Release: https://chiesirarediseases.com/media/european-commission-authorization-of-prx-102
Image Credits – Marie-Claire GUBLER, Paris, France, CC BY 2.0 https://creativecommons.org/licenses/by/2.0, via Wikimedia Commons
Recommended Companies
More Headlines