Valneva Stock Price Drops 40% as UK cancels vaccine deal
Valneva shares dropped by around 40% in early trading on Monday as the UK government announced its intention to withdraw from a deal with the French pharma company for 100 million doses of its yet-to-be-approved COVID-19 vaccine.
In a press release statement today, the company said:
“Valneva SE, a specialty vaccine company, today announced that it has received a termination notice from the UK government (HMG) in relation to the supply agreement for its Covid-19 vaccine candidate, VLA2001. The contract provides HMG with the right to terminate. HMG has alleged that the Company is in breach of its obligations under the Supply Agreement, but the Company strenuously denies this.”
The vaccine company “strenuously denies” a breach of its obligations under the agreement, but did not elaborate further on the details of the accused breach. Valneva continued to explain that it is moving ahead with its VLA2001 development plan. Vilneva’s pivotal Phase 3 trial, Cov-Compare, is currently ongoing at Public Health England (“PHE”).
The company’s vaccine manufacturing site is located in Livingston, west Scotland, which was visited by the British Prime Minister, Boris Johnson, in January this year.
Scotland’s health secretary, Humza Yousaf, commented for BBC Good Morning Scotland:
“This is a blow for the facility in Livingston. We are very keen and will be reaching out to the company to try to get security and secure a future for that facility in Livingston; we hope that would be with Valneva. Clearly, when it comes to their supposed alleged failure to meet their contract obligations, we obviously are looking for more information from the UK government and would expect that shortly.”

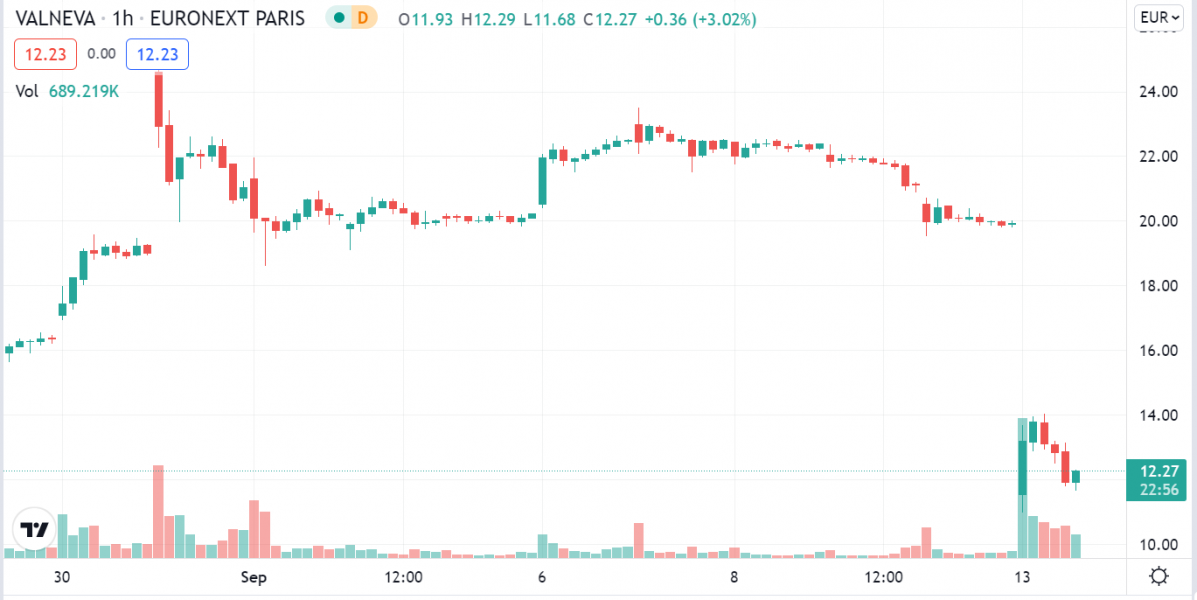
The announcement of the termination resulted in just over a 40% drop in Valneva’s Stock Price: VLA.PA (Paris) €11.88 -8.04(-40.36%) but has shown some signs of recovery to -38.50% at the time of writing.
Advertising
More Headlines