Measuring Plasma Protein Binding: The Key to Unlocking Drug Efficacy and Safety
Plasma protein binding is a phenomenon where drugs bind to proteins in the blood, thereby reducing the concentration of free, unbound drug in the bloodstream. Historically, the importance of plasma protein binding in drug discovery was not fully appreciated. However, in recent years, researchers have recognized that plasma proteins play a crucial role in controlling the free drug concentration in plasma and in compartments in equilibrium with plasma, which can effectively attenuate drug potency in vivo.
Sovicell, an innovative company that develops groundbreaking products for preclinical pharmaceutical research, has developed a unique plasma protein binding assay to help researchers accurately determine the unbound fraction of drugs that are tightly bound to plasma proteins – the TRANSIL High Sensitivity Binding Assay.
The significance of plasma protein binding for drug discovery
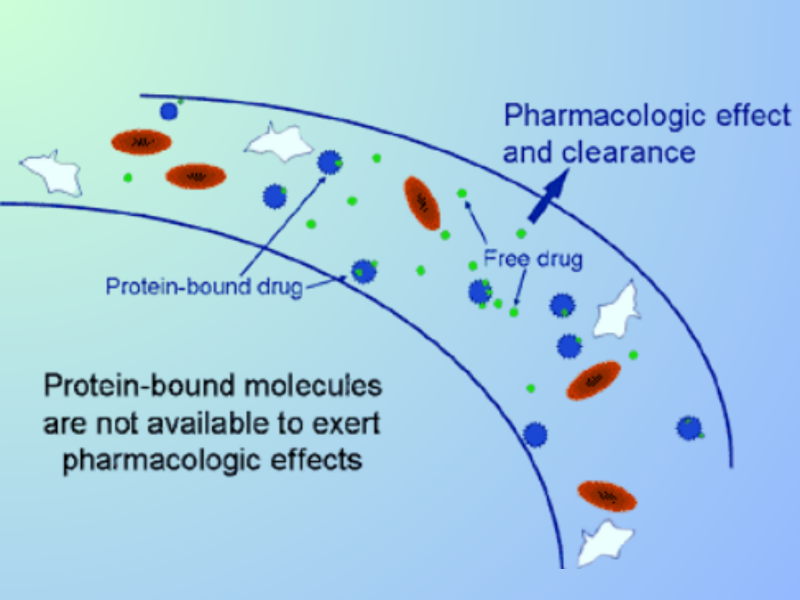
The implications of plasma protein binding on the pharmacokinetics of drugs have been well recognized for some time, but there is relatively little published literature on the effects of plasma protein binding on the availability of a drug at the site of action and on pharmacological action. Nevertheless, studies have shown that the binding of drugs to plasma proteins can significantly affect their pharmacokinetics and pharmacodynamics. The historical background and thermodynamic basis for the ‘Free Drug Principle’ have been extensively studied and are well understood.
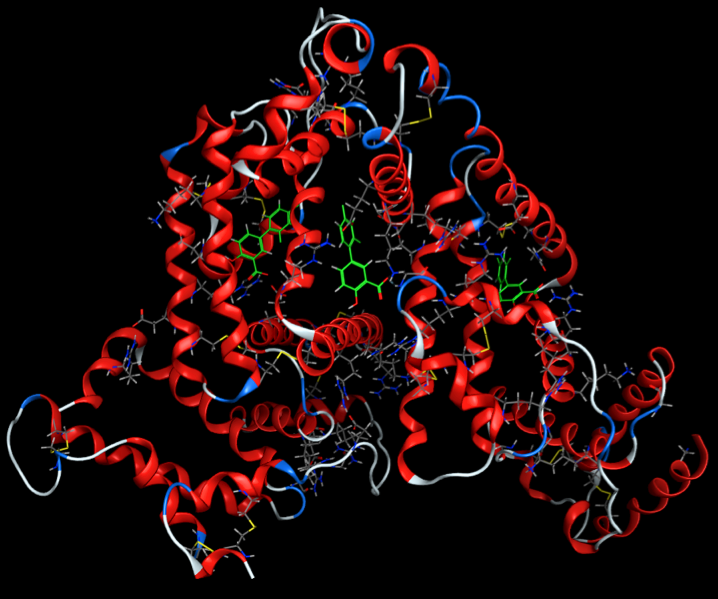
Two proteins are primarily responsible for plasma binding. They are serum albumin and α1-acid glycoprotein. Both are present in sufficient quantity and are capable of binding a broad variety of drugs with sufficient affinity to have a significant effect on drug action. Human serum albumin (HSA) is the major protein component of plasma, and it constitutes 4.5% of the weight of human blood. It is capable of binding endogenous ligands, such as fatty acids and metal ions, as well as drugs. There are multiple drug-binding sites on HSA, although two appear to predominate, and there is a slight preference for binding acidic drugs.
α1-Acid glycoprotein (AAG), on the other hand, is an acidic, heavily glycosylated protein. It consists of a single amino acid chain of 204 residues. AAG has been shown to exhibit a preference for binding basic and neutral drugs. There have been multiple drug-binding sites reported, but one site appears to be the most important, particularly for basic drugs.
Understanding plasma protein binding to improve drug efficacy and safety profiles
In the drug discovery process, plasma protein binding is an essential consideration when designing and developing new drugs. Plasma protein binding can significantly affect a drug’s pharmacokinetics, including its bioavailability, clearance, and distribution, which can ultimately affect its efficacy and safety profile. Researchers must determine the extent of plasma protein binding for a drug to assess its potency and potential adverse effects. By understanding the effects of plasma protein binding on drug action in vivo, researchers can design drugs that exhibit improved efficacy and safety profiles.
Plasma protein binding impacts drug efficacy and safety
Plasma protein binding is a crucial factor in drug discovery as it can significantly affect a drug’s pharmacokinetics and pharmacodynamics, ultimately impacting its efficacy and safety profile. Two plasma proteins, human serum albumin (HSA) and α1-acid glycoprotein (AAG), are present in sufficient quantity and have been extensively studied for their binding properties and their impact on drug action in vivo has been well established. By taking into account the effects of plasma protein binding during the drug discovery process, researchers can design drugs that exhibit improved efficacy and safety profiles.
Sovicell has designed a groundbreaking ready-to-use assay kit to address the challenges researchers face. Hinnerk Boriss, Chief Executive Officer of Sovicell explains:
Sovicell has developed a novel tool, the TRANSIL High Sensitivity Binding Assay, to address the challenges posed by highly bound, sticky, or hardly soluble drug candidates. This assay accurately determines the unbound fraction of drugs that are tightly bound to plasma proteins, even when the unbound fraction is below 0.001%, yielding high recovery for drugs that exhibit high unspecific binding or low solubility.
The innovative TRANSIL High Sensitivity Binding Assay Kit
To tackle the challenges posed by highly bound, sticky, or hardly soluble drug candidates, Sovicell’s TRANSIL High Sensitivity Binding Assay uses membrane coated beads in conjunction with plasma to measure the amount of free drug. One of the main advantages of this assay is its high sensitivity, which can detect low levels of free drug even when the drug candidate is highly bound to plasma proteins.

Highly lipophilic and sticky compounds also create notorious difficulties in plasma protein binding assays by critically reducing the recovery or erroneous results due to uneven non-specific binding or precipitation in the plasma and buffer compartments, or due to analytical challenges because of a very low remaining drug concentration.
The TRANSIL High Sensitivity Binding Kit circumvents these issues by having plasma in all assay compartments. Therefore, the TRANSIL High Sensitivity Binding Kit accurately determines the unbound fraction of drugs that are tightly bound to plasma proteins – even when the unbound fraction is below 0.001%. Additionally, the TRANSIL assay yields high recovery for drugs that exhibit too high unspecific binding in other assay systems, or precipitate because of low solubility. The assay kit indirectly determines the fraction of a drug bound to plasma by measuring the partitioning of drug between the plasma proteins and artificial cell membranes.
Related products: TRANSIL High Sensitivity Binding Assay