Transthyretin (TTR) for Neurodegenerative Disease Research

StressMarq has developed unique Transthyretin (TTR) Filaments and Monomers designed to propel research on TTR and its association with amyloid diseases.
TTR Filaments and Monomers
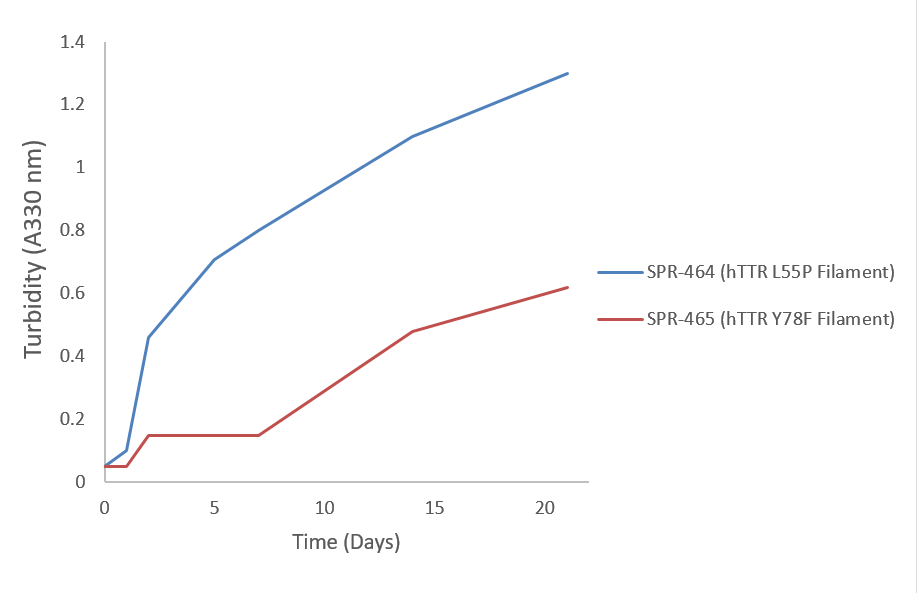
The misfolding of Transthyretin (TTR) is associated with Amyloid diseases. The L55P and Y78F mutants of TTR are known to be amyloidogenic, contributing to the development of these diseases. StressMarq offers both L55P and Y78F TTR filaments and monomers, providing researchers with valuable tools for studying these variants. TTR aggregation, a key aspect of amyloid diseases, can be quantitatively measured using turbidity as an indicator.
StressMarq’s Transthyretin L55P Variant Filaments and Transthyretin Y78F Variant Filaments are specialized research tools for investigating distinct mutations in the transthyretin (TTR) protein.
The L55P variant filaments allow researchers to study the unique behavior of the L55P tetramer, which can dissociate into monomeric amyloidogenic intermediates and form fibril precursors. The Transthyretin Y78F Variant Filaments focus on the Y78F mutant of TTR, enabling researchers to explore the effects of this mutation on TTR structure, stability, and fibrillogenesis.
These filaments are compatible with a range of applications including Western blot (WB), sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), in vivo assays, and in vitro assays, making them versatile tools for researchers delving into Amyotrophic Lateral Sclerosis (ALS) disease, Alzheimer’s disease, blood-related studies, cardiovascular system research, cell signaling, lipid and lipoprotein metabolism, metabolism, neurodegeneration, Parkinson’s disease, and tangles & tau.
These variant filaments together with their respective accompanying monomers (Transthyretin L55P or Y78F Variant Monomers) provide comprehensive tools for researchers aiming to understand the implications of specific mutations in TTR and their relevance in various diseases.

(Catalog No. SPR-464)
About Transthyretin (TTR)
Transthyretin (TTR) is a crucial transport protein found in the serum and cerebrospinal fluid, responsible for carrying the thyroid hormone thyroxine and retinol-binding protein bound to retinol. Its primary function is to maintain the balance and distribution of these essential molecules within the body. However, when TTR undergoes misfolding and aggregation, it becomes associated with amyloid diseases such as senile systemic amyloidosis (SSA), familial amyloid polyneuropathy (FAP), and familial amyloid cardiomyopathy (FAC). TTR also demonstrates potential beneficial effects by binding to beta-amyloid protein, which prevents the accumulation of beta-amyloid plaques typically associated with Alzheimer’s disease. This suggests that TTR may play a protective role in mitigating the development of Alzheimer’s pathology. Understanding the intricate properties and diverse implications of TTR is essential for elucidating the mechanisms underlying amyloid diseases and exploring potential therapeutic strategies.
For more information on Transthyretin (TTR) Filaments and Monomers for your research please contact StressMarq Biosciences today or browse the full catalog here: