SOD1 for Neurodegenerative Disease Research

StressMarq has developed unique Superoxide Dismutase 1 (SOD1) Pre-formed Fibrils (PFFs) and monomers for neurodegenerative disease research, particularly valuable for researchers in understanding the role of SOD1 in Amyotrophic Lateral Sclerosis (ALS).
SOD1 Pre-Formed Fibrils (PFFs)
SOD1 Pre-Formed Fibrils (PFFs) are synthetic fibrillar aggregates of SOD1 that can induce the aggregation of endogenous SOD1 in vitro and in vivo. They can be used as a tool to study the molecular mechanisms of SOD1 aggregation and toxicity, as well as to screen for potential therapeutic agents that can prevent or reverse SOD1 aggregation. SOD1 PFFs can also serve as a model for other protein aggregation diseases, such as Alzheimer’s disease and Parkinson’s disease.
StressMarq’s SOD1 Pre-Formed Fibrils (PFFs) are produced by incubating recombinant human SOD1 monomers at high temperature and agitation for several days. The resulting fibrils are purified by centrifugation and characterized by Thioflavin T fluorescence, transmission electron microscopy, and SDS-PAGE.
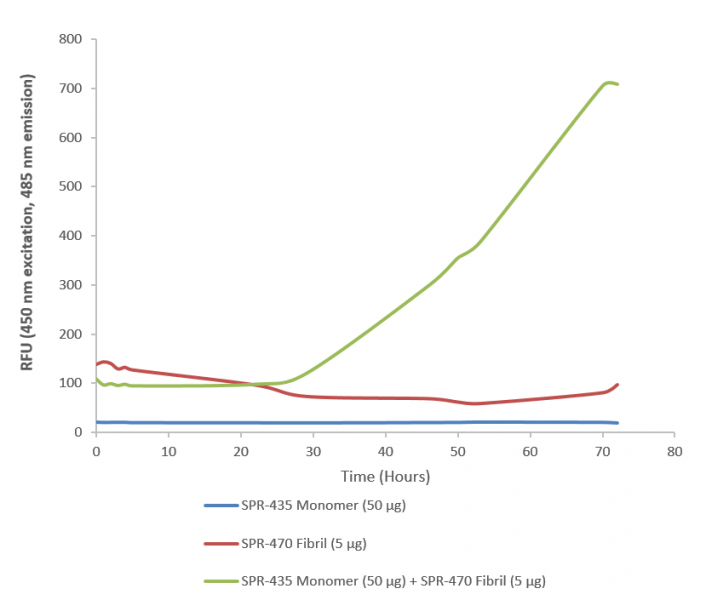
SOD1 PFFs seed the aggregation of SOD1 monomers in a concentration- and time-dependent manner, as demonstrated by increased Thioflavin T fluorescence in seeded aggregation assays.
The seeded aggregation of SOD1 monomers mimics the pathological process of SOD1 aggregation in ALS and can be used to investigate the factors that influence this process, such as pH, temperature, metal ions, or post-translational modifications.

Superoxide Dismutase 1 (SOD1)
SOD1 is a highly conserved and ubiquitously expressed enzyme that plays a vital role in the antioxidant defense system of cells. It catalyzes the dismutation of the harmful superoxide radical (O2•–) into less toxic oxygen (O2) and hydrogen peroxide (H2O2), which are then further metabolized by catalase and glutathione peroxidase. SOD1 contains copper (Cu) and zinc (Zn) ions that are essential for their catalytic activity and structural stability. The copper ion cycles between Cu2+ and Cu+ states during the reaction, while the zinc ion is not redox active but serves to stabilize the SOD1 structure. The two subunits of SOD1 form a homodimer of 32 kDa, linked by two cysteines forming an intra-subunit disulfide bridge.
Linking SOD1 Gene Mutations in ALS Pathogenesis
SOD1 misfolding and aggregation into neurotoxic species is implicated in Amyotrophic Lateral Sclerosis (ALS), a fatal neurodegenerative disorder that causes progressive loss of motor neurons and muscle function. ALS can be sporadic (sALS) or familial (fALS), with about 20% of fALS cases linked to mutations in the SOD1 gene. More than 150 mutations have been identified, most of them affecting the folding, stability, or interactions of SOD1. These mutations may result in increased oxidative stress, impaired axonal transport, mitochondrial dysfunction, inflammation, or impaired proteostasis. The exact mechanism by which SOD1 aggregates cause neuronal death is still unclear, but it may involve direct toxicity, prion-like propagation, or impairment of normal SOD1 function.
Understanding the Role of SOD1 in ALS Pathogenesis
StressMarq’s cutting-edge SOD1 Pre-Formed Fibrils (PFFs) are a valuable tool for neuroscience research, particularly important for investigating the role of SOD1 in the development of ALS and formulating innovative approaches for therapeutic strategies to advance drug discovery for neurodegenerative diseases. They can also be used to study the effects of SOD1 aggregation on cellular functions, such as oxidative stress response, mitochondrial dynamics, autophagy, or apoptosis. Furthermore, they can be used to test the efficacy and safety of potential drugs that target SOD1 aggregation or its downstream consequences.
For more information on SOD1 products please contact StressMarq Biosciences today or browse the full catalog here: