Tau Proteins for Neuroscience Research

StressMarq is a pioneer in the development of tau proteins that facilitate the investigation of tau aggregation, a key characteristic of neurodegenerative diseases like Alzheimer’s disease.
Active Tau Proteins for Neuroscience Research
Active tau proteins serve as invaluable tools in studying neurodegenerative diseases, particularly Alzheimer’s disease, where aberrant tau aggregation is a prominent feature. StressMarq leads the field in manufacturing active tau proteins, enabling researchers to delve into the intricate mechanisms underlying tau pathology.
StressMarq’s active tau proteins open up brand new avenues for investigating the molecular and cellular mechanisms underlying tau-associated neurodegenerative diseases, enhancing researchers understanding and paving the way for the development of novel treatment strategies.
Tau Pre-formed Fibrils (PFFs) for Neuroscience Research
StressMarq stands at the forefront as the pioneering provider of tau pre-formed fibrils (PFFs) and filaments, revolutionizing the field of neuroscience research. StressMarq is the first company to offer these essential tools, enabling neuroscientists and researchers to unravel the intricate complexities of tau pathology and its association with neurodegenerative disorders.
The novel tau pre-formed fibrils (PFFs) empower the investigation of tau aggregation in a controlled laboratory setting. Active tau PFFs act as seeding agents, recruiting tau monomers and facilitating their assembly into larger fibrillar structures, mimicking the pathological tau aggregates observed in Alzheimer’s disease and other tauopathies characterized by the deposition of abnormal tau protein in the brain.
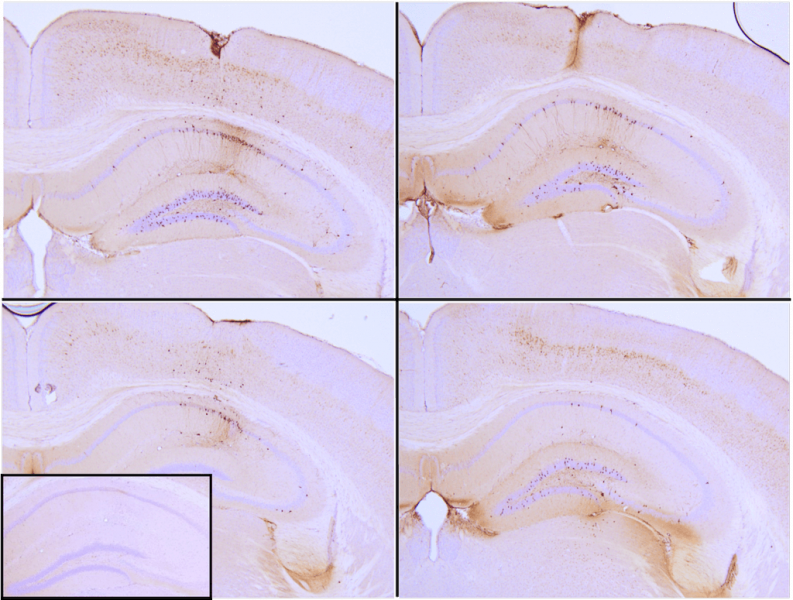
These active tau PFFs initiate the process of tau aggregation by recruiting monomers, resulting in the formation of larger tau fibrils. The efficacy of this process has been demonstrated through thioflavin T assays, which show an increase in fluorescence indicating tau fibrillization when active tau PFFs are combined with active tau monomers. Additionally, specific tau PFFs have been injected into P301L mice, effectively seeding tau aggregation and inducing tau pathology in the hippocampus. This approach enables researchers to examine the progression of neurodegeneration and evaluate potential therapeutic interventions.
Tau Monomers
StressMarq offers a comprehensive range of human and mouse tau monomers. The monomeric forms of tau proteins are crucial for studying their aggregation properties and the underlying mechanisms in neurodegenerative diseases. By offering monomeric tau proteins, StressMarq enables researchers to delve into the molecular and cellular processes involved in tau aggregation, providing valuable insights into neurodegenerative diseases and aiding in the development of potential therapeutic interventions.
Range of Tau PFFs and Monomers
StressMarq’s tau monomers and fibrils are available in full-length isoforms (2N4R or Tau-441) or truncated forms (K18). Tau-441 has a molecular weight of approximately 46 kDa, while K18 tau weighs around 15 kDa. Another fragment called dGAE, consisting of amino acids 297-391, is an essential component of paired helical filaments (PHFs) and assembles into PHF-like fibrils in vitro without the need for additives or templates.
The tau proteins are available in wild-type form or with various mutations. Mutations such as P301S, P301L, K280 deletion, and C322A are associated with frontotemporal dementia and impact tau’s assembly and fibrillization properties. Transgenic mouse models with P301S and P301L mutations are commonly used in tau research. The K280 deletion mutation promotes fibrillization into PHFs without the presence of heparin or other inducers, while the C322A mutation enhances the formation of PHFs.
Full-length PFFs have a higher effectiveness in seeding fibrillization, but a combination of both full-length and truncated forms can be particularly toxic to neurons. Most tau varieties are fibrillized using a heparin scaffold, while soluble tau filaments are formed using a linear anionic scaffold. Notably, dGAE tau and K18 K280 deletion tau can fibrillize without the need for scaffolds.
StressMarq offers the option to express tau PFFs using baculovirus in addition to E. coli. Baculovirus-derived tau PFFs undergo post-translational modifications, fibrillize without the need for a scaffold, and are endotoxin-free, ensuring their suitability for research purposes.
For more information about the range of tau proteins and range of PFF’s and monomers please contact StressMarq Biosciences today or browse the full catalog here: